The primary goal of the Moore lab is to understand the molecular mechanisms by which neuronal activity instructs circuit formation and function in the mammalian brain.
Prior to any external cues, the basic neuronal circuitry is hardwired in early development through molecular programs and spontaneous activity. With the onset of sensory experiences and throughout adulthood, fluctuations in activity levels work to shape and refine immature networks and adapt to perturbations. Homeostatic plasticity mechanisms work to maintain a set level of activity through modulation of synaptic strength and membrane excitability in individual neurons. Such plasticity mechanisms are induced when neuronal excitation activates intracellular signaling pathways to initiate new gene expression. Therefore, genes responsive to changes in neuronal activity via calcium influx are ideal candidates for translating experience-dependent changes in the neuron. Despite the recognized importance of activity-dependent mechanisms in shaping circuit function, the genetic underpinnings of these processes are poorly understood.
The Moore lab seeks to understand how a neuron can sense and respond to its environment to alter the brain’s output. We use a combination of techniques including electrophysiology, mouse genetics, molecular biology, and behavioral assays to seek to understand how intrinsic plasticity mechanisms shape neuron function and behavior. Toward this goal we seek to address the following questions.
1) What are the molecular mechanisms that instruct changes in neuronal intrinsic excitability?
From crustaceans to mammals, neurons respond to decreased activity by modulating their ionic conductance in order to re-establish their appropriate firing rate, via a homeostatic adjustment of intrinsic excitability. How these functional changes are instructed at the molecular level remain largely unknown. To address this question, we employ a systematic approach to screen candidate genes using whole-cell recordings and imaging techniques.
2) How are different plasticity mechanisms regulated?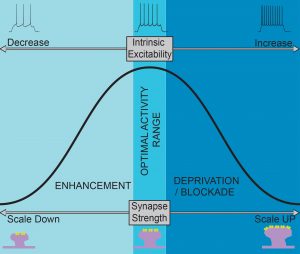
In response to altered activity a neuron can adapt by adjusting either its synaptic inputs or its intrinsic properties and include both Hebbian and homeostatic plasticity. However, it remains unclear how different plasticity mechanisms are engaged at the molecular level to induce cell specific changes in neural function.
3) What are the functional consequences of impaired intrinsic excitability on circuit plasticity?
One outstanding question, which we seek to address, is what happens when an individual neuron is unable to adjust its basal firing rate in response to altered activity. Or how removing the neuron’s ability to adapt shapes brain behavior. We combine electrophysiological recordings, mouse genetics and behavioral techniques to investigate the underlying contribution of intrinsic plasticity to circuit function.